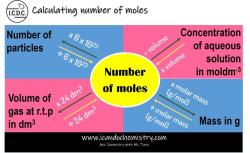
What are the concentration units of mole fraction and molality?
Mole Fraction and Molality are two important concentration units in chemistry, and they are used to describe the concentration of solute in a solution. Here's an explanation of each:
1. Mole Fraction (X):
- Definition: Mole fraction is a dimensionless ratio that expresses the ratio of the number of moles of solute to the total number of moles in a solution.
- Formula: Mole Fraction (X) of a component A in a solution is calculated using the following formula:X(A) = (moles of A) / (total moles in the solution)
- Range: Mole fractions range from 0 to 1, with 0 indicating no presence of the component in the solution, and 1 indicating that the entire solution consists of that component.
- Usage: Mole fraction is particularly useful in colligative properties of solutions and in cases where a precise measure of the relative amount of a substance in a solution is required.
2. Molality (m):
- Definition: Molality is a measure of the concentration of a solute in a solution, and it relates the amount of solute to the mass of the solvent.
- Formula: Molality (m) is calculated using the following formula:m = (moles of solute) / (mass of solvent in kg)
- Units: Molality is typically expressed in moles of solute per kilogram of solvent, which makes it different from molarity, which is expressed in moles of solute per liter of solution.
- Usage: Molality is particularly useful in certain chemical reactions and colligative properties (like boiling point elevation and freezing point depression) because it's not dependent on the temperature or the density of the solution. It remains constant regardless of temperature.
In summary, mole fraction is a dimensionless ratio that describes the relative number of moles of a component in a solution, while molality is a measure of the concentration of a solute in a solution relative to the mass of the solvent. Both are valuable tools in chemistry for describing and calculating the concentration of solute in solutions, and they have specific applications in different contexts.
What are the concentration units of mole fraction and molality in chemistry?
Mole fraction is a unit of concentration that is expressed as the ratio of the moles of a specific component to the total moles of all components in a solution. It is a dimensionless quantity, which means that it does not have any units.
Molality is a unit of concentration that is expressed as the number of moles of a solute dissolved in one kilogram of solvent. It is typically expressed in units of mol/kg.
How do you calculate and interpret mole fraction and molality?
Mole fraction can be calculated using the following equation:
Mole fraction of component A = moles of component A / total moles of all components
Molality can be calculated using the following equation:
Molality of solute B = moles of solute B / mass of solvent in kilograms
Once you have calculated the mole fraction or molality of a solution, you can interpret it as follows:
- Mole fraction: The mole fraction of a component in a solution represents the proportion of that component in the solution. For example, a mole fraction of 0.5 for component A means that half of the molecules in the solution are component A.
- Molality: The molality of a solution represents the concentration of the solute in the solution relative to the mass of the solvent. For example, a molality of 1 mol/kg for solute B means that there is one mole of solute B dissolved in every kilogram of solvent.
Can you provide examples of using mole fraction and molality in chemical solutions?
Here are some examples of using mole fraction and molality in chemical solutions:
- Mole fraction:
- A solution of ethanol and water with a mole fraction of ethanol of 0.5 is known as a 50% ethanol solution.
- A solution of salt (NaCl) and water with a mole fraction of salt of 0.01 is known as a 1% saline solution.
- Molality:
- A solution of sulfuric acid (H2SO4) in water with a molality of 1 mol/kg is known as a 1 molal sulfuric acid solution.
- A solution of sodium hydroxide (NaOH) in water with a molality of 10 mol/kg is known as a 10 molal sodium hydroxide solution.
What are the advantages of using these concentration units in specific scenarios?
Mole fraction and molality are useful concentration units in specific scenarios because they can be used to express the concentration of a solution in a way that is independent of temperature and volume. This is important for some applications, such as thermodynamics and chemical kinetics.
Mole fraction is also a useful concentration unit for describing solutions of non-electrolytes, which are substances that do not dissociate into ions when dissolved in water.
Molality is a useful concentration unit for describing solutions of electrolytes, which are substances that do dissociate into ions when dissolved in water. This is because molality is not affected by the dissociation of electrolytes.
How to convert between different concentration units in chemistry?
There are a number of ways to convert between different concentration units in chemistry. One common method is to use the following equation:
concentration in unit A = concentration in unit B * conversion factor
The conversion factor will depend on the two concentration units that you are converting between. For example, the conversion factor to convert from mole fraction to molality is:
conversion factor = 1000 g/kg
This is because molality is defined as the number of moles of solute dissolved in one kilogram of solvent.
Here is an example of how to convert from mole fraction to molality:
A solution of ethanol and water has a mole fraction of ethanol of 0.5. What is the molality of the ethanol in the solution?
conversion factor = 1000 g/kg
molality of ethanol = mole fraction of ethanol * conversion factor
molality of ethanol = 0.5 * 1000 g/kg
molality of ethanol = 500 mol/kg
You can also use online calculators or conversion tables to convert between different concentration units.