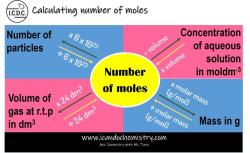
How do you find molarity from density?
To find molarity from density in a chemistry context, you need additional information, specifically the mass of the solute and the molecular weight of the solute. The formula to calculate molarity (M) from density (ρ), mass (m), and molecular weight (Mw) is as follows:
Where:
- is the molarity of the solution (in moles per liter, mol/L or M).
- is the mass of the solute (in grams, g).
- is the density of the solution (in grams per milliliter, g/mL).
- is the molecular weight of the solute (in grams per mole, g/mol).
Here are the steps to find molarity from density:
Determine the Mass of the Solute (m):
- You need to know the mass of the solute in the solution. This can be obtained through measurements or from the information provided.
Find the Molecular Weight of the Solute (Mw):
- The molecular weight is the sum of the atomic weights of all the atoms in a molecule. You can find the molecular weight of the solute using a periodic table and adding up the atomic weights of its constituent atoms.
Determine the Density of the Solution (ρ):
- The density of the solution is usually provided, or you can measure it using a graduated cylinder or similar equipment.
Apply the Formula:
- Plug the values you've gathered into the formula:
- Ensure that the units for mass, density, and molecular weight are compatible. For example, if you use grams for mass and g/mL for density, the resulting molarity will be in moles per liter (M or mol/L).
Calculate the Molarity (M):
- Perform the calculation to find the molarity of the solution.
This formula is particularly useful when you know the mass of a solute and the volume of the solution, but you want to express the concentration in molarity. Remember to double-check your units to ensure consistency in your calculations.
How to calculate molarity from density
To calculate molarity from density, you can use the following formula:
molarity = (density * percent concentration) / (molar mass * 100)
where:
- molarity is in units of moles per liter (mol/L)
- density is in units of grams per milliliter (g/mL)
- percent concentration is the percentage of the solute in the solution
- molar mass is the mass in grams of one mole of the solute
Relationship between molarity and density in chemistry
Molarity and density are two different ways of measuring the concentration of a solution. Molarity is the number of moles of solute per liter of solution, while density is the mass of solution per unit volume.
There is a relationship between molarity and density because the mass of a solution is equal to the mass of the solute plus the mass of the solvent. The mass of the solvent is constant, so the mass of the solution is directly proportional to the mass of the solute.
This means that the density of a solution will increase as the molarity of the solution increases. However, the relationship is not linear, and the density of a solution will also depend on the molar masses of the solute and solvent.
Formula or equation for converting density to molarity
The following formula can be used to convert density to molarity:
molarity = (density * percent concentration) / (molar mass * 100)
where:
- molarity is in units of moles per liter (mol/L)
- density is in units of grams per milliliter (g/mL)
- percent concentration is the percentage of the solute in the solution
- molar mass is the mass in grams of one mole of the solute
How to determine the concentration of a substance using its density
To determine the concentration of a substance using its density, you can use the following steps:
- Determine the density of the solution.
- Determine the percent concentration of the solute in the solution.
- Determine the molar mass of the solute.
- Use the formula above to calculate the molarity of the solution.
Online calculators or tools to assist in molarity calculations
There are a number of online calculators and tools that can assist in molarity calculations. One popular example is the molarity calculator on the Thermo Fisher Scientific website.
This calculator allows you to enter the density, percent concentration, and molar mass of the solute to calculate the molarity of the solution. The calculator also allows you to enter the molarity and density of the solution to calculate the percent concentration or molar mass of the solute.