Is mg L equal to ppm?
Milligrams per liter (mg/L) and parts per million (ppm) are often used interchangeably to express the concentration of a substance in a solution. However, they are not precisely equal because they represent slightly different ways of expressing the same concentration.
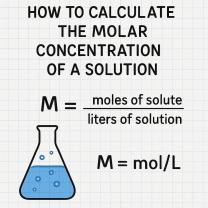
Milligrams per Liter (mg/L): This unit of measurement indicates the mass of a substance (in milligrams) dissolved in one liter of a solution. It is a measure of concentration based on the mass of the solute (substance being dissolved) per unit volume of the solution.
Parts Per Million (ppm): Parts per million is a dimensionless unit that expresses the ratio of the number of parts of a substance to one million parts of the entire solution. In other words, it represents the number of parts of the solute per one million parts of the solution.
To understand the relationship between mg/L and ppm, consider the following:
1 mg/L is equivalent to 1 ppm when the density of the solution is very close to 1 g/mL (grams per milliliter). This is often the case for water, where the density is approximately 1 g/mL.
For water-based solutions, when you have 1 mg of a substance dissolved in 1 liter of water (1 mg/L), it is also equivalent to 1 ppm. This is because 1 mg of solute in 1 liter of water is a concentration of 1 part per million.
However, if the density of the solution deviates significantly from 1 g/mL, the relationship between mg/L and ppm can differ slightly. To calculate ppm in such cases, you may need to consider the density of the solution.
In practical terms, for most everyday purposes and when dealing with water-based solutions, mg/L and ppm are often used interchangeably because the difference is negligible. However, in scientific and analytical contexts where precision is crucial, it's essential to account for the density of the solution to ensure accurate conversions between mg/L and ppm.
Milligrams per Liter (mg/L) and Parts Per Million (ppm): Equivalency Explained
Milligrams per liter (mg/L) and parts per million (ppm) are both units of concentration that are used to measure the amount of a substance dissolved in a solution.
One ppm is equivalent to one milligram of a substance dissolved in one liter of solution. In other words, 1 ppm = 1 mg/L.
This equivalency is only true for dilute solutions, where the density of the solution is approximately equal to the density of water. For more concentrated solutions, the density of the solution may be significantly different from the density of water, in which case the equivalency between ppm and mg/L will not be exact.
Converting mg/L to ppm: A Practical Guide
To convert mg/L to ppm, simply divide the concentration in mg/L by 1000. For example, a concentration of 10 mg/L is equivalent to 10 ppm.
Here is a simple formula for converting mg/L to ppm:
ppm = mg/L / 1000
Interpreting Concentration Units: The Relationship Between mg/L and ppm
Both mg/L and ppm are used to measure the concentration of a substance in a solution, but they are different units. mg/L is a mass-to-volume unit, while ppm is a ratio of parts.
This means that mg/L measures the amount of a substance in milligrams per liter of solution, while ppm measures the number of parts of a substance per million parts of solution.
For example, a concentration of 1 mg/L of chlorine in water means that there is 1 milligram of chlorine dissolved in every liter of water. A concentration of 1 ppm of chlorine in water means that there is 1 part of chlorine per million parts of water.
In other words, 1 ppm is equivalent to one part per million, which is a very small amount.
Conclusion
mg/L and ppm are both important units of concentration that are used in a variety of applications. By understanding the relationship between these two units, you can easily convert between them and interpret concentration data accurately.