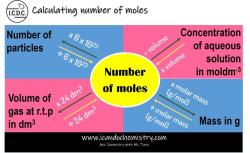
Is molarity the same as concentration?
Molarity and concentration are related concepts, but they are not exactly the same. They both describe the amount of a solute (substance being dissolved) in a solution (a homogeneous mixture of a solvent and a solute), but they use slightly different ways of expressing this information.
Here's the key difference:
Molarity (M): Molarity is a measure of the concentration of a solute in a solution and is defined as the number of moles of solute per liter of solution. It is expressed in moles per liter (mol/L or M). The formula for calculating molarity is:
Molarity (M) = Moles of Solute (mol) / Volume of Solution (L)
Molarity is particularly useful in chemistry when you need to know the precise number of moles of solute in a given volume of solution.
Concentration: Concentration is a more general term that refers to the amount of solute in a given amount of solution. It can be expressed in various units, depending on the context and the field of science. Some common units of concentration include mass/volume (e.g., grams per liter), volume/volume (e.g., milliliters per liter), and percentage by mass or volume.
Mass/Volume Concentration: This is often expressed in grams per liter (g/L) or milligrams per milliliter (mg/mL). For example, if you have 10 grams of sugar dissolved in 1 liter of water, the mass/volume concentration of sugar is 10 g/L.
Volume/Volume Concentration: This is expressed in milliliters per liter (mL/L). For example, if you mix 100 mL of ethanol with 900 mL of water, the volume/volume concentration of ethanol is 100 mL/L.
Percentage Concentration: This is expressed as a percentage of the solute's amount relative to the total solution. For example, if you dissolve 20 grams of salt in 100 grams of water, the percentage concentration of salt is 20%.
In summary, molarity is a specific and standardized way to express concentration, where concentration is a broader term that encompasses various units and methods for expressing the amount of solute in a solution. Molarity is commonly used in chemistry for its precision and utility in stoichiometry and chemical reactions, while other concentration units may be more appropriate in different scientific fields or practical applications.
Molarity vs. Concentration: Understanding the Difference
Concentration is a general term that refers to the amount of a substance dissolved in a solution. It can be expressed in a variety of ways, such as grams per liter (g/L), moles per liter (M), or percentage.
Molarity is a specific type of concentration that is expressed in moles per liter (M). It is the number of moles of a solute dissolved in one liter of solution.
Exploring the Distinction Between Molarity and Concentration
The main distinction between molarity and concentration is that molarity is a specific type of concentration that is expressed in moles per liter. Concentration is a more general term that can be expressed in a variety of ways.
For example, a solution of 100 grams of NaCl dissolved in 1 liter of water would have the following concentration:
- Concentration in g/L: 100 g/L
- Concentration in M: 1.69 M (calculated by dividing the grams of NaCl by its molar mass)
- Concentration in percentage: 10% (calculated by dividing the grams of NaCl by the total grams of solution and multiplying by 100)
As you can see, the concentration of the solution can be expressed in a variety of ways. However, molarity is the most specific way to express concentration, as it takes into account the number of moles of solute in solution.
Molarity and Concentration: Are They Truly Equivalent?
Molarity and concentration are not truly equivalent. Molarity is a specific type of concentration that is expressed in moles per liter. Concentration is a more general term that can be expressed in a variety of ways.
Molarity is the most specific way to express concentration, as it takes into account the number of moles of solute in solution. This is important for many chemical calculations, such as determining the amount of a reactant needed to produce a certain amount of product.
However, there are some cases where it is more convenient to express concentration in other units. For example, the concentration of salt water is often expressed in grams per liter (g/L), as this is a more familiar unit to most people.
Conclusion
Molarity and concentration are two different ways to express the amount of a substance dissolved in a solution. Molarity is a more specific type of concentration that is expressed in moles per liter. Concentration is a more general term that can be expressed in a variety of ways.
Molarity is the most useful way to express concentration for many chemical calculations. However, there are some cases where it is more convenient to express concentration in other units.