How much sodium bicarbonate to raise pH?
The amount of sodium bicarbonate (baking soda) you need to raise the pH of a solution depends on several factors, including the initial pH of the solution, the volume of the solution, and the desired final pH. Sodium bicarbonate is a weak base and can be used to increase pH. Here are some general guidelines and measurements to help you adjust the pH using sodium bicarbonate:
Determine the Initial and Desired pH:First, measure the initial pH of the solution you want to adjust. Then, determine the desired final pH that you want to achieve. This will help you calculate how much sodium bicarbonate is needed.
Calculate the pH Difference:Find the difference between the desired pH and the initial pH. For example, if the initial pH is 5.0, and you want to raise it to 7.0, the pH difference is 2.0.
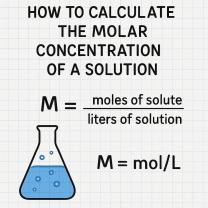
Determine the Volume of the Solution:Measure or estimate the volume of the solution you want to treat. You'll need this information to calculate the amount of sodium bicarbonate required.
Consult a pH Adjustment Table:Sodium bicarbonate pH adjustment tables or charts are available to provide approximate measurements for how much sodium bicarbonate is needed to achieve a specific pH change in a given volume of solution. These tables take into account the solution's buffering capacity.
Use the Henderson-Hasselbalch Equation (Optional):For more precise calculations, you can use the Henderson-Hasselbalch equation, which takes into account the pKa of the acid and the concentration of the acid and its conjugate base. The equation is as follows:
Where:
- pH is the desired pH.
- pKa is the negative logarithm (base 10) of the acid dissociation constant of the weak acid.
- [A^-] is the concentration of the conjugate base (e.g., from sodium bicarbonate).
- [HA] is the concentration of the weak acid in the solution.
Gradually Add Sodium Bicarbonate:Based on the information from the pH adjustment table or your calculations, add the appropriate amount of sodium bicarbonate to the solution. It's important to add it gradually and stir thoroughly to ensure even distribution.
Monitor pH and Adjust as Needed:After adding the sodium bicarbonate, monitor the pH of the solution. It may take some time for the pH to stabilize. If the pH is still not at the desired level, you can make further adjustments by adding more sodium bicarbonate.
Exercise Caution:Sodium bicarbonate is a weak base, and while it's generally safe to use for small pH adjustments, be cautious not to over-adjust the pH, especially in situations where precise pH control is critical. Over-adjustment can lead to imbalances and unintended consequences in the solution.
Remember that the specific measurements and calculations will depend on the unique characteristics of your solution, so it's important to consult appropriate references or seek advice from a chemist or expert in the field if you're working with complex or critical pH adjustments.
Sodium bicarbonate, commonly known as baking soda, is a versatile compound with various applications, including pH adjustment. Let's explore its use in pH control:
Using Sodium Bicarbonate to Adjust pH Levels
Sodium bicarbonate is a mild alkali, meaning it can neutralize acids and raise the pH of a solution. It's often used to adjust pH in various contexts, such as:
Pool Maintenance: Adding baking soda to pool water can raise the pH, making it less acidic and more comfortable for swimmers.
Aquarium Care: Maintaining a stable pH in aquariums is crucial for aquatic life. Baking soda can be used to raise the pH if it becomes too acidic.
Food Preparation: Baking soda is used in baking to react with acidic ingredients, producing carbon dioxide that helps baked goods rise.
How to Calculate the Amount of Sodium Bicarbonate Needed
The amount of baking soda required to adjust pH depends on the initial pH, the desired pH, and the volume of the solution. A general formula is:
Amount of NaHCO3 (grams) = (Volume of solution (liters) * (Desired pH - Initial pH)) / 84
For example, to raise the pH of 1 liter of solution from 5 to 7, you would need:
Amount of NaHCO3 = (1 * (7 - 5)) / 84 = 0.0238 grams
Risks and Precautions When Using Sodium Bicarbonate for pH Adjustment
While baking soda is generally safe, precautions are necessary:
Gradual Addition: Add baking soda slowly and in small increments to avoid overshooting the desired pH.
Monitoring pH: Regularly monitor the pH using a pH meter or test strips to ensure it stays within the desired range.
Avoid Excess: Excessive baking soda can cause alkalosis, an imbalance in body chemistry, if ingested in large quantities.
Alternative pH Adjustment Methods
Other methods for adjusting pH include:
Acids: Acids like hydrochloric acid or citric acid can lower pH. Use caution when handling strong acids.
Buffers: Buffers, such as phosphate buffers, can maintain a stable pH within a specific range.
pH Control Systems: Automated pH control systems can continuously monitor and adjust pH using pumps and sensors.
Real-World Applications of Sodium Bicarbonate for pH Control
Sodium bicarbonate is widely used for pH control in various fields:
Wastewater Treatment: Baking soda is used to neutralize acidic wastewater before discharge to protect aquatic ecosystems.
Agriculture: Soil pH can be adjusted with baking soda to optimize nutrient availability for crops.
Pharmaceuticals: Baking soda is used in antacids to neutralize stomach acid and relieve heartburn.
Sodium bicarbonate is a valuable tool for pH adjustment, with applications ranging from everyday household tasks to industrial processes.