What is a doubt or uncertainty in measurement?
In the context of measurement and metrology, doubt or uncertainty refers to the lack of complete confidence in the precision or exactness of a measured value or result. It represents the range within which the true value of the quantity being measured is likely to lie, taking into account various sources of error and variability in the measurement process.
Measurement uncertainty is an inherent aspect of all measurements because no measurement process can be perfectly precise or accurate. Several factors contribute to measurement uncertainty, including:
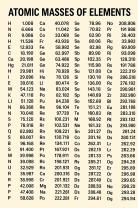
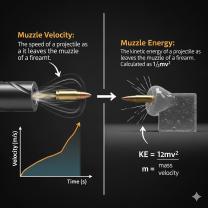
Instrument Precision: The limitations of the measuring instrument itself, such as the smallest division on a ruler or the resolution of a digital sensor.
Systematic Errors: Systematic errors are consistent biases or inaccuracies introduced by the measurement system or method. They can be caused by calibration issues, instrument drift, or imperfect measurement techniques.
Random Errors: Random errors are unpredictable variations in measurements that occur due to factors such as environmental conditions, operator technique, or inherent variability in the quantity being measured.
Reproducibility: The degree to which measurements can be consistently reproduced by different operators or using different instruments.
Bias: Bias refers to a consistent deviation of measurements from the true value. It can result from systematic errors and calibration issues.
Resolution: The smallest change in the quantity being measured that the instrument can detect.
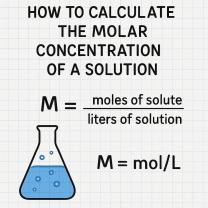
Measurement uncertainty is typically expressed with a numerical value and a confidence interval. For example, a measurement might be reported as "10.5 ± 0.2 cm," indicating that the true value of the quantity is likely to be between 10.3 cm and 10.7 cm with a high level of confidence.
Understanding and quantifying measurement uncertainty is essential in various fields, including science, engineering, manufacturing, and quality control. It helps ensure that measurement results are reliable, and it allows for informed decision-making. Techniques for estimating and managing measurement uncertainty include statistical analysis, calibration, and quality control procedures.
Accurate and precise measurement, along with a well-characterized measurement uncertainty, is critical in fields where safety, compliance with standards, or the validity of scientific experiments and research depend on the reliability of measurements.
Defining Doubt and Uncertainty in the Context of Measurement
Doubt in the context of measurement refers to the lack of confidence in the result of a measurement. It can be caused by a variety of factors, such as the limitations of the measuring instrument, the skill of the person performing the measurement, and the environmental conditions in which the measurement is performed.
Uncertainty in the context of measurement refers to the range of values within which the true value of the measurand is likely to lie. It is typically expressed as a standard deviation or a confidence interval.
Uncertainty is an important concept in measurement because it allows us to quantify the reliability of our results. It is also important to consider uncertainty when making decisions based on measurement results. For example, if we are measuring the amount of a drug in a sample, the uncertainty in the measurement will affect our ability to determine the appropriate dosage for a patient.
Uncertainty in Scientific and Industrial Measurements
Uncertainty is present in all measurements, but it is especially important to consider in scientific and industrial measurements, where high levels of accuracy and precision are required.
In scientific research, uncertainty can be caused by a variety of factors, such as the limitations of the experimental apparatus, the purity of the chemicals used, and the skill of the researcher.
In industrial settings, uncertainty can be caused by factors such as the accuracy of the measuring instruments, the stability of the environment, and the variability of the product being measured.
Uncertainty Analysis: Essential Concepts in Measurement
Uncertainty analysis is a process for quantifying and understanding the uncertainty in measurement results. It is an essential tool for scientists and engineers who need to make reliable and accurate measurements.
There are a number of different approaches to uncertainty analysis, but they all involve the following steps:
- Identify the sources of uncertainty. What factors could affect the accuracy and precision of the measurement?
- Quantify the uncertainty from each source. This may involve statistical analysis, experimental data, or expert judgment.
- Combine the individual uncertainties to get an overall uncertainty estimate.
- Report the uncertainty along with the measurement result. This allows others to understand the reliability of the result and make informed decisions based on it.
Uncertainty analysis is a complex topic, but it is essential for anyone who needs to make reliable and accurate measurements. There are a number of resources available to help scientists and engineers learn more about uncertainty analysis and how to apply it to their work.