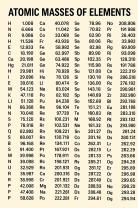
What is 1 part per million?
One part per million (1 ppm) is a unit of measurement that represents a very small quantity or concentration of a substance in relation to a larger quantity. It is commonly used in various fields, including chemistry, environmental science, and manufacturing, to express the proportion of one substance (the "part") in one million parts of a mixture or solution.
Mathematically, 1 ppm is equivalent to 1 part of a substance per 1,000,000 parts of the whole. This can be expressed as a fraction or as a decimal:
- As a fraction: 1 ppm = 1/1,000,000
- As a decimal: 1 ppm = 0.000001
To put this into perspective, imagine you have a million identical marbles, and you want to find out how many of them are of a particular color. If only one of those marbles is the desired color, you would say that the concentration of that color in the entire collection is 1 ppm.
1 ppm is an extremely small measurement and is often used when dealing with trace amounts of substances or pollutants in air, water, soil, or other materials. It's a useful way to quantify very low concentrations, especially when assessing the potential environmental or health impact of substances.
What Is 1 Part Per Million (PPM)? Exploring the Concept
One part per million (PPM) is a unit of measurement that is used to describe very small concentrations of a substance in a larger solution. It is equivalent to one part in a million parts. PPM is a very precise unit of measurement, and it is often used in scientific and engineering applications.
The Basics of PPM: Defining a Fundamental Unit of Measurement
PPM is a dimensionless unit of measurement, meaning that it does not have a unit of its own. It is simply a ratio of one part to a million parts. PPM can be used to measure the concentration of any substance in any solution, regardless of the units of measurement used for the substance or the solution.
PPM Simplified: Understanding the Value of One Part Per Million
One PPM is a very small concentration. To put it in perspective, one PPM is equivalent to one drop of water in an Olympic-sized swimming pool. It is also equivalent to one grain of sand on a beach that stretches from New York City to San Francisco.
Applications of PPM
PPM is used in a wide variety of applications, including:
- Environmental science: PPM is used to measure the concentration of pollutants in air, water, and soil.
- Food safety: PPM is used to measure the concentration of pesticides, herbicides, and other chemicals in food.
- Industrial hygiene: PPM is used to measure the concentration of hazardous chemicals in the workplace.
- Manufacturing: PPM is used to measure the purity of materials and products.
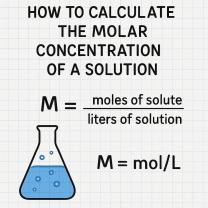
How to Calculate PPM
To calculate PPM, simply divide the mass or volume of the solute by the mass or volume of the solution, then multiply by a million. For example, if you have 1 milligram of salt dissolved in 1 liter of water, the concentration of salt in the solution would be 1 PPM (1 mg / 1 L * 1 million = 1 PPM).
Conclusion
PPM is a very useful unit of measurement for describing very small concentrations of a substance in a larger solution. It is used in a wide variety of applications, including environmental science, food safety, industrial hygiene, and manufacturing.