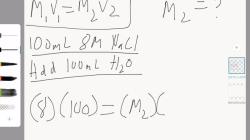How do you calculate volume when given mass?
To calculate volume when given mass, you need to know the density of the material. The formula connecting mass, volume, and density is:
Rearranging the formula, you can solve for volume:
For example, if you have the mass of a substance (let's say it's grams) and you know the density of that substance (let's say it's grams per cubic centimeter), you can calculate the volume using the formula:
This means the volume of the substance with a mass of grams and a density of grams per cubic centimeter is cubic centimeters.
What's the method to calculate volume based on given mass?
There are two main methods to calculate volume based on a given mass:
1. Using density: This is the most common and straightforward method. It relies on the formula:
Volume = Mass / Density
where:
- Volume (V) is the quantity you're trying to find, measured in cubic units (cm³, mL, etc.).
- Mass (m) is the given mass of the object, measured in grams (g), kilograms (kg), etc.
- Density (ρ) is the intrinsic property of the material, indicating its "compactness," measured in grams per cubic centimeter (g/cm³), kilograms per cubic meter (kg/m³), etc.
Steps:
- Identify the mass (m) of the object.
- Determine the density (ρ) of the material. This can be found in tables, databases, or by conducting experiments.
- Substitute the values of m and ρ into the formula.
- Calculate the volume (V).
Example:
- Mass (m) = 50 grams (g)
- Density (ρ) of iron = 7.87 g/cm³
V = 50 g / 7.87 g/cm³ ≈ 6.36 cm³
Therefore, the volume of the iron object is approximately 6.36 cubic centimeters.
2. Using geometric formulas: This method applies when you know the object's specific shape and dimensions. Different shapes have their own volume formulas, like cubes, spheres, cylinders, etc. You'll need to measure the relevant dimensions (length, width, height, radius) and plug them into the appropriate formula along with the mass and density (if needed).
This method is more complex but can be necessary for irregularly shaped objects where density might not be readily available.
Let me know if you'd like to explore specific examples or have any further questions about volume calculations!